It was announced today that Titan Pharmaceutical’s new drug Probuphine was accepted for review by the FDA. What is Probuphine exactly? Probuphine is a six-month sub dermal implant containing buprenorphine hydrochloride (suboxone.) The drug has been developed as a way to deliver continuous long-term treatment for opioid addiction.
According to recent numbers, there are 2.2 million people suffering from opioid addiction in the United States. Approximately 20 percent of that 2.2 million are addicted to street drugs, such as heroin, and the other 80 percent to prescription drugs, such as oxycodone, hydrocodone, methadone, hydromorphone and codeine. Before 2000, medication assisted therapies for opioid addiction was limited to a select number of facilities around the US. The US Drug Addiction Treatment Act of 2000 changed all that by allowing medical office-based treatment of addiction, expanding access to medication-assisted therapies. After that, sales of buprenorphine (suboxone) went up; today they make about 1.75 billion dollars in the US alone.
Probuphine Under Review by FDA
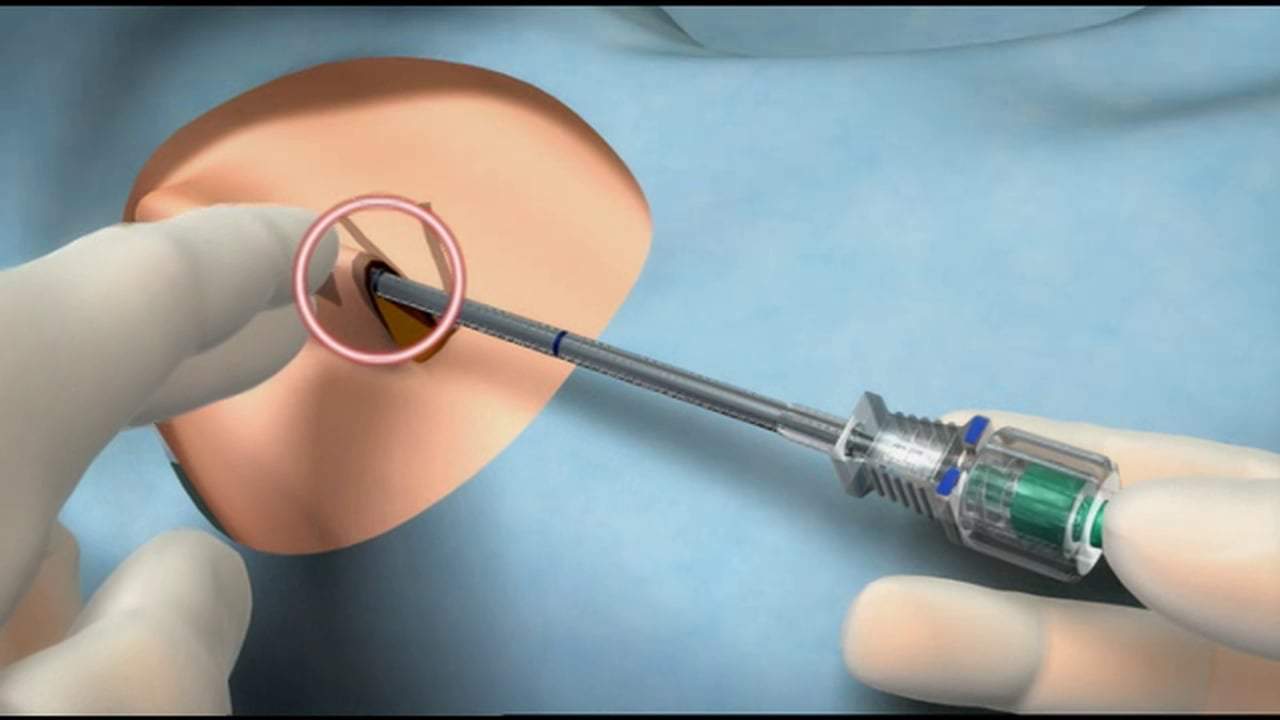
Probuphine is still in the investigational phase, according to the FDA, which means it is still being determined whether or not it is effective or safe for the general public. Basically, Probuphine goes under the skin (sub dermal) and is designed to deliver the medication buprenorphine for six months after a single treatment. This is meant to promote patient compliance.
Buprenorphine, which is the active ingredient in the medication, is a FDA approved drug for treatment of opiate addiction, and right now it is available in table and film form. Both of these forms require the patient to take them daily in order to be effective.
Probuphine was developed to combat this. Its delivery system consists of a small, solid, implant that can be placed under the skin in the upper arm of the patient. From that point forward the implant will deliver buprenorphine for the next 3 to 6 months before being taken out and then replaced for another six months if necessary.
The idea is that if a patient doesn’t have to remember to take a pill everyday they will be more stable, and at less risk of not taking their medication and relapsing. This implant will quite literally make buprenorphine a part of them so as to keep them from being able of not taking their medication on purpose or by it slipping their mind.
We will keep you update if this makes any further movements towards approval.
What do you think? Is sub dermal suboxone that lasts 6 months at a time a good idea or a bad idea? What do you think about the current long term medication assisted therapy system?






